A team of researchers from the Netherlands and Australia has developed a groundbreaking chemical network that emulates decision-making capabilities typically found in living organisms and computers. This innovative system, known as a recursive enzymatic competition network (ERN), showcases the ability to adapt and respond to external environmental signals, marking a significant advancement in the field of synthetic biology.
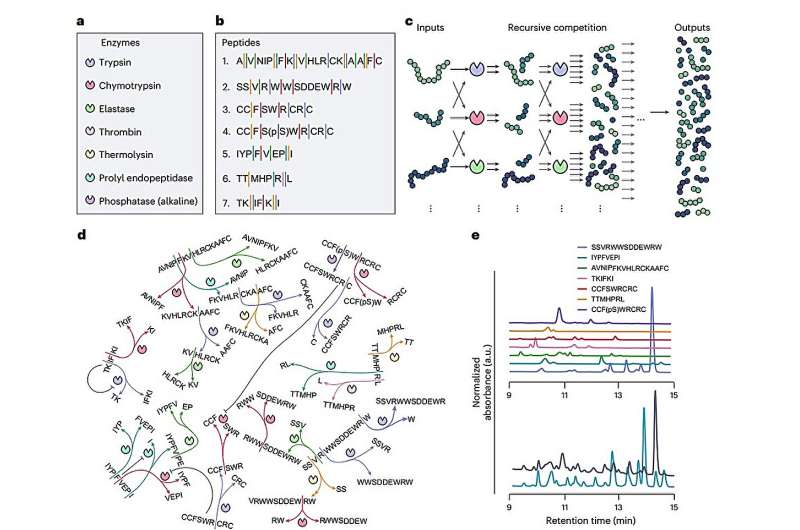
The findings, published in the journal Nature Chemistry, reveal that the ERN comprises seven enzymes and seven peptides that compete for proteases. This competition enables the chemical mixture to reorganize itself, allowing it to classify both chemical and physical signals. The network can accurately sense temperature variations between 25–55°C with a precision of approximately 1.3°C, and it can adjust its responses in real-time, similar to neural systems.
The ability to respond to environmental changes was once thought to be exclusive to complex living organisms. Traditional computers have long been designed to handle stimulus-response tasks, but replicating this behavior in chemical systems posed a challenge. The researchers utilized recursive interactions, where products of chemical reactions are fed back into the system for further processing, to create a highly dynamic environment capable of multitasking.
The ERN operates by continuously cutting and competing peptides, resulting in a constantly changing mixture of chemical fragments. This dynamic process generates a nonlinear network of enzymatic reactions, allowing the composition of the mixture to vary significantly based on initial inputs, such as peptide concentration and environmental factors like temperature or pH. Researchers measure these chemical fragments in real-time using a mass spectrometer, which provides data interpreted by a straightforward algorithm known as a linear readout layer. This layer decodes fragment patterns, translating them into predictions or decisions, such as sensing temperature changes or detecting periodic light pulses.
The implications of the ERN extend beyond academic interest. The researchers believe that the capabilities demonstrated by this system could lead to the development of smarter, more adaptive biosensors and materials. Potential applications encompass fields such as healthcare and technology, where enhanced sensing and information processing could revolutionize current methodologies.
This research underscores a significant step towards mimicking the complexities of biological systems, which constantly monitor their environments to make informed decisions. While previous attempts to design synthetic networks have struggled to capture the full intricacies of living systems, the ERN represents a leap forward by employing recursive interactions and competitive dynamics.
In summary, the development of this enzyme network marks a pivotal moment in chemical systems, demonstrating that decision-making processes can be replicated outside of traditional living organisms. The research, led by Souvik Ghosh and his team, opens the door to innovative applications and deeper understanding of how chemical networks can function autonomously in response to their surroundings.