On February 19, 2026, Dr. Ali May, an assistant professor at the Icahn School of Medicine at Mount Sinai, delivered a compelling presentation on her research regarding the role of tissue-resident macrophages in the development of embryonic salivary glands. This talk was part of the Department of Biology Seminar Series and highlighted how macrophages influence organogenesis beyond their traditional functions of immune response and tissue maintenance.
Dr. May challenged the conventional perception of macrophages as mere “garbage collectors,” tasked with engulfing dead cells and debris. Instead, she urged attendees to recognize these cells as vital players in developmental processes, capable of interacting with epithelial cells, nerves, and other supportive tissues. Her insights stem from her postdoctoral work on neurons, where she first observed the intersection of immune cells and the nervous system during development.
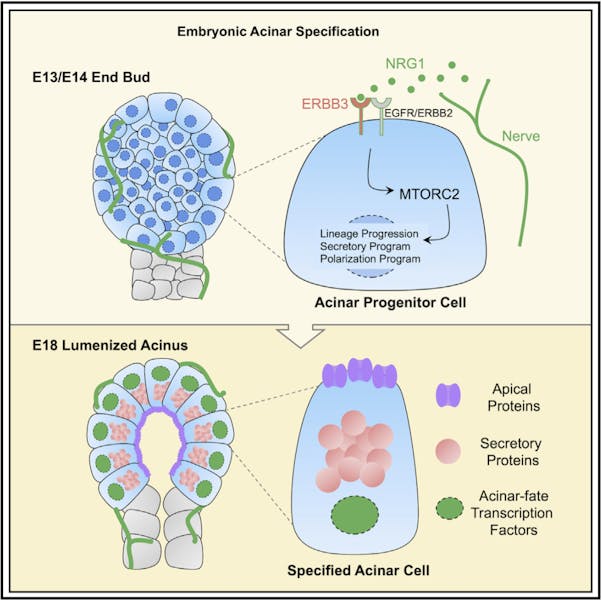
The research team employed a range of advanced methodologies, including live imaging, genetic depletion, and organ culture techniques, to explore how varying macrophage populations affect branching morphogenesis—the process by which glands develop their tree-like structures. A key aspect of their approach involved an inducible genetic system that selectively eliminates macrophages during critical windows of salivary gland development. After observing the effects of macrophage depletion, the team noted a significant reduction in the branching of salivary glands.
Dr. May explained, “When you have less branching, you’re going to have much larger buds and fewer of them.” The study revealed that embryos deprived of macrophages exhibited abnormal structures known as ‘Terminal Buds,’ indicating impaired branching morphogenesis. Gene expression analysis further corroborated these findings, showing decreased levels of genes linked to the differentiation of secretory acinar cells and other epithelial markers.
The absence of macrophages not only stunted branching but also negatively impacted the maturation of essential cell types responsible for saliva production. “Not only are we reducing branching,” Dr. May emphasized, “but we’re hindering differentiation of these essential cells.” Moreover, the study uncovered developmental repercussions on non-epithelial cell types, suggesting the presence of a distinct nervous system within the salivary gland. This observation aligns with the hypothesis that macrophages play a role in pruning neurons during development in various organs.
Transitioning to the topic of signaling, Dr. May explained her focus on ligand-receptor interactions. To identify potential communication pathways, her lab analyzed single-cell RNA sequencing data, which highlighted tumor necrosis factor (TNF) as a significant factor expressed in macrophages. Traditionally associated with inflammation, TNF had not been regarded as a key player in developmental regulation.
Dr. May noted, “We generally think of TNF as a pro-inflammatory marker in an injury or a disease state within the adult organ.” However, tissue staining confirmed TNF expression in macrophages adjacent to the epithelial compartment, raising intriguing questions about its developmental role. The research team observed a marked increase in the expression of TNF receptors in epithelial cells during development, suggesting that TNF signaling may contribute to the maturation of salivary glands.
To assess the impact of TNF on epithelial development directly, Dr. May’s team utilized an in-vitro culture system. They isolated the epithelial components of embryonic salivary glands, excluding nerves, blood vessels, and macrophages, to create a purified environment for observation. “You can see them undergoing branching and then the formation of buds at the ends of these branches,” she described, likening them to “little spiders in dishes” as they developed.
Remarkably, when TNF was added to the culture, the glands exhibited enhanced growth and branching. “We see a significant increase in both the branching and the growth of these glands,” she reported, noting the visible formation of ducts within the tissues, which she described as “strong dark lines in the center of the branches.”
Dr. May concluded her presentation by discussing the dual origins of macrophages in salivary glands: a long-lived yolk-sac-derived population and a later-arriving monocyte-derived population. These distinct subtypes appear to serve different functions throughout development. Notably, she showcased live imaging footage illustrating macrophages crossing the basement membrane to envelop the epithelial compartment, a behavior she termed “cuddling.”
“What we can see is that this population can actually cross the basement membrane and enter into the epithelial compartment,” she explained. This interaction raises important questions about whether macrophages are simply monitoring dividing cells or actively stimulating their proliferation. The findings suggest that macrophages are integral to the dynamics of epithelial growth, challenging the long-held view of their passive role.
Dr. May’s research opens new avenues for understanding the complex interplay between immune cells and organ development, emphasizing the importance of macrophages in shaping not only structural but also functional aspects of embryonic glands. The implications of her work extend beyond salivary glands, potentially informing broader insights into developmental biology and organogenesis.