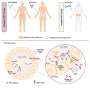
Polycystic kidney disease (PKD) is a hereditary condition characterized by the development of fluid-filled cysts in the kidneys. These cysts can lead to significant pain and loss of organ function, often resulting in the need for dialysis in advanced stages. Currently, there is no cure for this debilitating disease. Researchers at UC Santa Barbara have proposed a novel therapy aimed at halting the aggressive growth of these cysts by utilizing monoclonal antibodies designed for targeted treatment.
Innovative Approach to Treatment
The research team, led by Thomas Weimbs, has developed a cyst-targeted therapy that could mitigate the relentless growth of PKD cysts. Their findings were published in Cell Reports Medicine, where they emphasize the need for a drug that can effectively penetrate the cysts and inhibit their proliferation. “The cysts just keep growing endlessly,” Weimbs stated, highlighting the urgency of their work.
Current treatment options primarily consist of small-molecule drugs, but these often come with significant side effects and toxicity, particularly affecting surrounding kidney tissues. The only drug currently available that shows some efficacy in slowing the disease’s progression is limited in its therapeutic application due to these adverse effects. The research team identified a promising alternative in the form of dimeric immunoglobulin A (dIgA), a monoclonal antibody capable of crossing epithelial membranes.
Mechanism of Action and Research Findings
The study builds on previous work published in 2015 that explored the potential of dIgA to target specific receptors within kidney cysts. Unlike traditional immunoglobulin G (IgG) antibodies, which are too large to access cyst interiors, dIgA can navigate through epithelial cell layers. This ability allows the antibody to enter the cysts and target the cMET receptor, a key driver of cyst growth.
After engineering the dIgA antibody, the researchers conducted tests on mouse models. They confirmed that the antibody successfully infiltrated the cysts and effectively inhibited cMET activity, leading to a reduction in cell growth signals. Notably, the treatment triggered a significant onset of apoptosis (cell death) in cyst epithelial cells while sparing healthy renal tissue, indicating a targeted therapeutic effect.
Despite the promising results, Weimbs cautioned that the research is still in preclinical stages. He noted that there are several challenges ahead, including the need for collaboration with partners interested in PKD and access to facilities capable of producing the necessary antibodies.
The team also plans to explore additional growth factors and receptors that may be involved in cyst progression, which could enhance the effectiveness of their approach. “In the literature, there are dozens of growth factors that have been shown to be active in these cyst fluids,” Weimbs explained. The next steps will involve comparing various antibodies and possibly combining them to achieve optimal results in slowing or reversing the disease’s effects.
The work conducted by Weimbs and his team, including lead author Margaret F. Schimmel and other collaborators, represents a significant advancement in the quest for effective treatments for PKD. As research continues, there is hope that this innovative approach could one day lead to a viable therapeutic option for those impacted by this challenging condition.
For more detailed information, refer to the study, “Development of a cyst-targeted therapy for polycystic kidney disease using an antagonistic dimeric IgA monoclonal antibody against cMET,” published in Cell Reports Medicine in 2025.