Research conducted by scientists at the University of Manchester has uncovered significant insights into how the immune system of the gut reacts after a stroke and its potential role in gastrointestinal complications. This study, published in the journal Brain, Behavior and Immunity, expands on the concept of the “gut-brain axis,” suggesting a communicative relationship between the gut and brain that influences health and disease.
The findings are particularly relevant given that stroke, a serious medical emergency characterized by disrupted blood flow to the brain, can lead to lasting impacts on both mobility and cognitive function. Patients often experience secondary bacterial infections and gastrointestinal issues, including difficulty swallowing and constipation. Researchers are increasingly recognizing that these complications may stem from alterations in the commensal microbiota, the beneficial bacteria that maintain gut health.
The study highlights a gap in understanding the mechanisms behind these gastrointestinal symptoms and their implications for stroke severity and recovery. Previous research by some of the study’s authors indicated that signals from the nervous system might modify gut immune responses post-stroke. The current research reveals that the gut-brain axis may operate bidirectionally, with immune cells that produce antibodies migrating to the brain and its surrounding membranes during a stroke, although the significance of this movement for stroke outcomes remains to be determined.
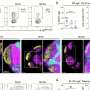
In their investigation, the researchers focused on mice to assess changes occurring in the small intestine following a stroke. They discovered that populations of immune cells responsible for antibody production were altered within days of the event. Notably, a specialized subset of cells producing Immunoglobulin A (IgA) was found to be hyper-activated. IgA plays a crucial role in regulating the populations of beneficial bacteria in the gut and is essential for maintaining gut health.
The team also observed that mice lacking IgA did not show the same degree of alterations in their gut microbiome after a stroke, indicating that changes in immune function might partially explain the gastrointestinal disturbances observed in stroke patients.
Lead investigator, Professor Matt Hepworth from the Lydia Becker Institute of Immunity and Inflammation at the University of Manchester, emphasized the complexity of stroke’s effects: “Stroke is a devastating neurological event but also has many long-term consequences that can leave the patient at risk of airway infection, as well as gastrointestinal complications.” He added that collaboration with neuroscientists has provided new insights into how gut immunity becomes disrupted after a stroke, potentially leading to changes in the management of beneficial gut bacteria.
Looking forward, Professor Hepworth noted the importance of understanding these immune changes: “While the focus remains on stroke prevention and early intervention to minimize damage in patients who suffer a stroke, we reveal a new understanding of the secondary pathologies that can affect recovery.” He highlighted the potential for immune-targeting therapeutics to address symptoms driven by immune responses following a stroke, which could ultimately enhance patients’ quality of life.
The study, titled “Cerebral ischaemic stroke results in altered mucosal antibody responses and host-commensal microbiota interactions,” was authored by Madeleine Hurry and colleagues, and is set to appear in the 2026 edition of the journal. The findings contribute to the growing body of evidence that underscores the intricate connections between the gut and brain, particularly in the context of stroke and its aftermath.