Vanderbilt Researchers Develop Nanobodies That Illuminate Toxic Alzheimer’s Proteins
In a groundbreaking advance, Vanderbilt Health and Vanderbilt University scientists have engineered tiny antibody fragments known as nanobodies that can cross the blood-brain barrier to “light up” and neutralize toxic amyloid protein aggregates linked to Alzheimer’s disease.
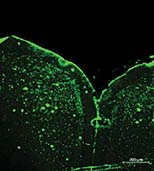
This breakthrough, reported in recent research, reveals how these fluorescently labeled nanobodies target soluble amyloid-beta oligomers (SAβOs) in the synapses—micro gaps between nerve cells critical for brain communication. These SAβOs are believed to disrupt memory and learning by attacking synapses before larger amyloid plaques form, pointing to a new frontier in early detection and treatment.
Nanobodies Target Alzheimer’s at Its Earliest Stage in the Brain
The team led by Wellington Pham, PhD, professor of Radiology and Biomedical Engineering, alongside Brian Wadzinski, PhD, and Ben Spiller, PhD, both associate professors of Pharmacology, genetically engineered nanobodies derived from unique antibodies found in alpacas living in Humphreys County, Tennessee. These tiny fragments specifically bind to SAβOs in the hippocampus, the brain’s memory center.
When injected intravenously, the nanobodies emit a fluorescent signal pinpointing toxic aggregates in mouse models of Alzheimer’s, enabling “precision in vivo molecular fingerprinting” that could revolutionize early diagnosis. More notably, one nanobody demonstrated the ability to neutralize these toxic aggregates, suggesting both diagnostic and therapeutic promise.
Global Potential: Australian Company Licenses Nanobody Technology
The innovation has attracted global attention, with an Australian biotechnology company licensing the technology to advance its development for clinical use. The company aims to explore the “theragnostic” potential—combining therapy and diagnostics—to detect and intervene in Alzheimer’s at its earliest, most treatable stages.
Collaboration and Funding Powering Alzheimer’s Breakthroughs
This pivotal research, initiated during the height of the COVID-19 pandemic in 2020, exemplifies multidisciplinary collaboration. Vanderbilt’s Imaging Science Institute, Brain Institute, Ingram Cancer Center, and Clinical and Translational Research Institute provided vital support. The project also received funding from the National Institute on Aging under grant R01AG061138.
“Nanobodies are a powerful tool to investigate synaptic dysfunction mechanisms that lead to neuronal death and tissue loss in Alzheimer’s,” said Dr. Pham.
Why This Discovery Matters Now
Alzheimer’s disease affects millions nationwide, and current diagnostics typically identify symptoms after irreversible brain damage occurs. The ability of Vanderbilt’s nanobodies to cross the blood-brain barrier and detect—and even neutralize—early toxic protein aggregates signals a seismic shift in managing this devastating disease.
By illuminating the earliest synaptic disruptions, these nanobodies could enable clinicians to diagnose Alzheimer’s before major cognitive decline, opening the door for treatments that slow or halt progression. For millions of Americans and countless families, this development offers a beacon of hope for earlier intervention and better outcomes.
What’s Next in Alzheimer’s Nanobody Research
Researchers are accelerating efforts to translate these findings into human trials and clinical tools. The licensed biotechnology company is expected to spearhead this next phase, aiming to refine the fluorescent nanobody probes and develop targeted therapies. Meanwhile, Vanderbilt continues to explore the molecular underpinnings of Alzheimer’s synaptic dysfunction to identify additional targets.
With over 6 million Americans living with Alzheimer’s and numbers rising, Vanderbilt’s nanobody innovation represents a critical new weapon in the fight to defeat this relentless disease.
For North Carolinians and US readers, this research underscores how cutting-edge science from a leading academic health system is forging new paths against Alzheimer’s, carrying profound implications for diagnosis, treatment, and the future of brain health nationwide.